In general, an atom’s electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. For this purpose, a dimensionless quantity, the Pauling scale, symbol χ, is the most commonly used. ElectronegativityĮlectronegativity, symbol χ, is a chemical property that describes the tendency of an atom to attract electrons towards this atom. The electron affinities of the noble gases have not been conclusively measured, so they may or may not have slightly negative values. Chlorine most strongly attracts extra electrons. Nonmetals like to gain electrons to form anions to have a fully stable electron shell. Nonmetals: Generally, nonmetals have more positive electron affinity than metals.Mercury most weakly attracts an extra electron. The electron affinity of metals is lower than that of nonmetals. Metals: Metals like to lose valence electrons to form cations to have a fully stable shell.It is well known that noble gases have closed electronic shell structure and hence have high ionization potentials and low electron affinities, due to which they are chemically inert and resistant to salt formation under most conditions.Īffinities of Nonmetals vs. Superhalogens are molecules that have electron affinities (EA) greater than that of Cl, the element with the highest EA (3.62 eV). In fact, the electron affinity of Cl, 3.62 eV is the largest of all the elements. Halogens have the highest electron affinities among all elements. This affinity is known as the second electron affinity, and these energies are positive. However, more energy is required to add an electron to a negative ion which overwhelms any release of energy from the electron attachment process. By convention, the negative sign shows a release of energy. This affinity is known as the first electron affinity, and these energies are negative. When an electron is added to a neutral atom, energy is released. To use electron affinities properly, it is essential to keep track of signs. Molecules with high electron affinity form very stable negative ions which are important in the chemical and health industry as they purify the air, lift mood, and most importantly, act as strong oxidizing agents. Na + e – → Na – – ∆H = Affinity = 52.8 kJ/molĮlectron affinity is one of the most important parameters that guide chemical reactivity. Electron affinities are more difficult to measure than ionization energies.Īn atom of Sodium in the gas phase, for example, gives off energy when it gains an electron to form an ion of Sodium. Note that ionization energies measure the tendency of a neutral atom to resist the loss of electrons. In other words, it can be expressed as the neutral atom’s likelihood of gaining an electron. The change in energy (in kJ/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:

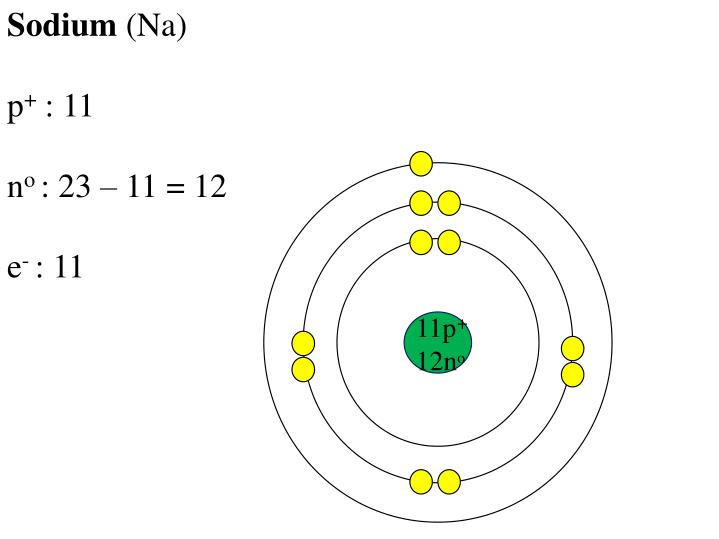
Electron Affinity and Electronegativity of SodiumĮlectron Affinity of Sodium is 52.8 kJ/mol.įirst Ionization Energy of Sodium is 5.1391 eV.
